Reaction mechanisms and reactive intermediates
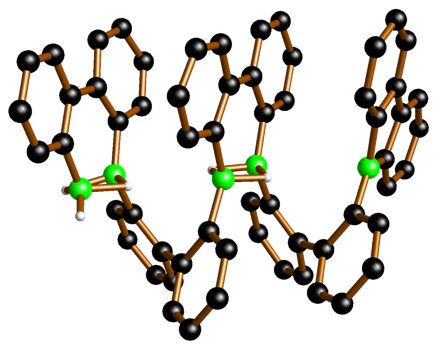 Dynamic covalent chemistry (DCC), the making and breaking of bonds within an equilibrating mixture of compounds, is a phenomenon often encountered when dealing with organoboranes and oligosilanes. Most often it is regarded as a nuisance, because it frequently leads to undesired substituent redistribution reactions. Yet, under carefully controlled conditions, dynamic covalent chemistry can be turned into a powerful synthetic tool, provided that it is possible to gain a detailed picture of the underlying mechanistic scenario. In the past few years, we have exploited DCC for the targeted condensation polymerization of ferrocenylboranes, the ring-opening oligomerization of 9H-9-borafluorene (cf. Figure), and the assembly of oligosilane chains, rings, and cages. In all cases, we have elucidated the reaction mechanisms by experimental as well as computational studies and identified the most relevant reaction intermediates.
Dynamic covalent chemistry (DCC), the making and breaking of bonds within an equilibrating mixture of compounds, is a phenomenon often encountered when dealing with organoboranes and oligosilanes. Most often it is regarded as a nuisance, because it frequently leads to undesired substituent redistribution reactions. Yet, under carefully controlled conditions, dynamic covalent chemistry can be turned into a powerful synthetic tool, provided that it is possible to gain a detailed picture of the underlying mechanistic scenario. In the past few years, we have exploited DCC for the targeted condensation polymerization of ferrocenylboranes, the ring-opening oligomerization of 9H-9-borafluorene (cf. Figure), and the assembly of oligosilane chains, rings, and cages. In all cases, we have elucidated the reaction mechanisms by experimental as well as computational studies and identified the most relevant reaction intermediates.
Molecules with unusual bonding situations
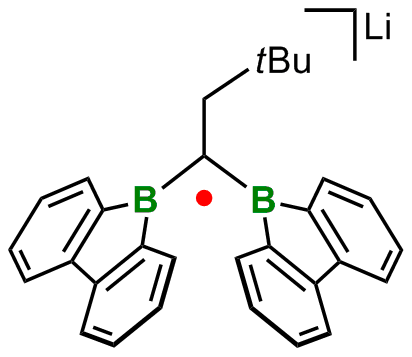 Pushing the limits of molecular stability is not only a rewarding intellectual challenge, but contributes decisively to a thorough understanding of chemical bonding. For example, our group succeeded in the synthesis and full characterization of species containing B•B one-electron two-center bonds. This bonding motif was previously known mainly from the elusive H2+ ion, which is extensively discussed in any textbook on quantum chemistry. Our research thus allowed for the first time to scrutinize the plethora of theoretical claims and predictions by solid experimental data.
Pushing the limits of molecular stability is not only a rewarding intellectual challenge, but contributes decisively to a thorough understanding of chemical bonding. For example, our group succeeded in the synthesis and full characterization of species containing B•B one-electron two-center bonds. This bonding motif was previously known mainly from the elusive H2+ ion, which is extensively discussed in any textbook on quantum chemistry. Our research thus allowed for the first time to scrutinize the plethora of theoretical claims and predictions by solid experimental data.
Homogeneous catalysis with transition metals or Frustrated Lewis Pairs (FLPs)
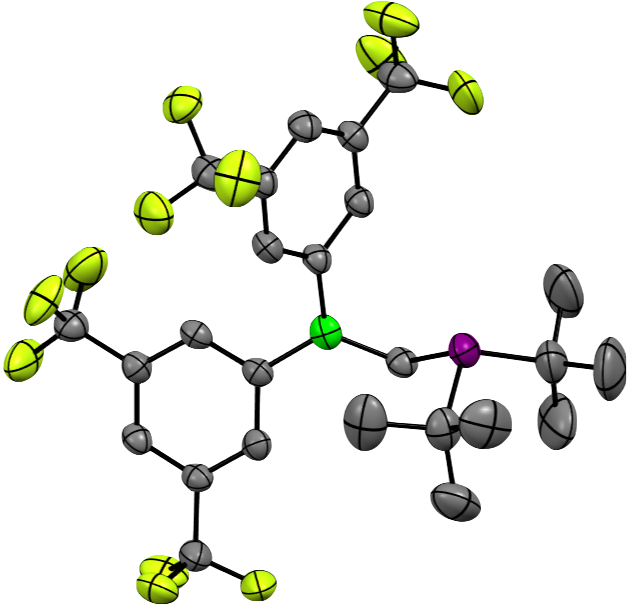 Homogeneous catalysts are essential tools in today’s synthesis laboratories. This has long been true for metal-based catalysts, and more recently also for “Frustrated Lewis Pairs (FLPs)”. Our research on transition metal catalysts focuses on the optimization of metal complexes for the derivatization of boron-containing organic materials, while leaving the B−C bonds intact. With regard to FLP development, we achieved the synthesis of geminal boron/phosphorous FLPs (cf. Figure). These molecules readily react with a broad selection of compounds. Apart from standard FLP substrates, such as H2, CO2 or imines, we also succeeded in the activation of ketones and alkyl halides.
Homogeneous catalysts are essential tools in today’s synthesis laboratories. This has long been true for metal-based catalysts, and more recently also for “Frustrated Lewis Pairs (FLPs)”. Our research on transition metal catalysts focuses on the optimization of metal complexes for the derivatization of boron-containing organic materials, while leaving the B−C bonds intact. With regard to FLP development, we achieved the synthesis of geminal boron/phosphorous FLPs (cf. Figure). These molecules readily react with a broad selection of compounds. Apart from standard FLP substrates, such as H2, CO2 or imines, we also succeeded in the activation of ketones and alkyl halides.
Organic electronics and luminescent materials
 Polycyclic aromatic hydrocarbons (PAHs) are highly promising optoelectronic materials. Key parameters, such as the electron affinity or the fluorescence wavelength, can be adjusted through the introduction of substituents at the perimeter of the PAH. An even greater effect is created when selected carbon atoms within the PAH scaffold are replaced by other p-block atoms. Our group is particularly interested in boron as the electronically perturbative element. As a rule of thumb, boron doping leads to the lowering of the LUMO energy level of a PAH, thereby providing access to excellent electron acceptors, which also tend to be strongly luminescent in the visible range of the electronic spectrum. The actual preparation of boron-containing PAHs is still a considerable challenge. In the recent past, we have developed synthesis routes based on B−B-coupling reactions, photocyclisation reactions, and the transition-metal mediated late-stage derivatisation of boron heterocycles. This enables us to systematically modify the molecular structure of a given target molecule, which is the prerequisite for an understanding of structure-property relationships.
Polycyclic aromatic hydrocarbons (PAHs) are highly promising optoelectronic materials. Key parameters, such as the electron affinity or the fluorescence wavelength, can be adjusted through the introduction of substituents at the perimeter of the PAH. An even greater effect is created when selected carbon atoms within the PAH scaffold are replaced by other p-block atoms. Our group is particularly interested in boron as the electronically perturbative element. As a rule of thumb, boron doping leads to the lowering of the LUMO energy level of a PAH, thereby providing access to excellent electron acceptors, which also tend to be strongly luminescent in the visible range of the electronic spectrum. The actual preparation of boron-containing PAHs is still a considerable challenge. In the recent past, we have developed synthesis routes based on B−B-coupling reactions, photocyclisation reactions, and the transition-metal mediated late-stage derivatisation of boron heterocycles. This enables us to systematically modify the molecular structure of a given target molecule, which is the prerequisite for an understanding of structure-property relationships.
Oligosilanes/-germanes and Si/Ge nanostructures:
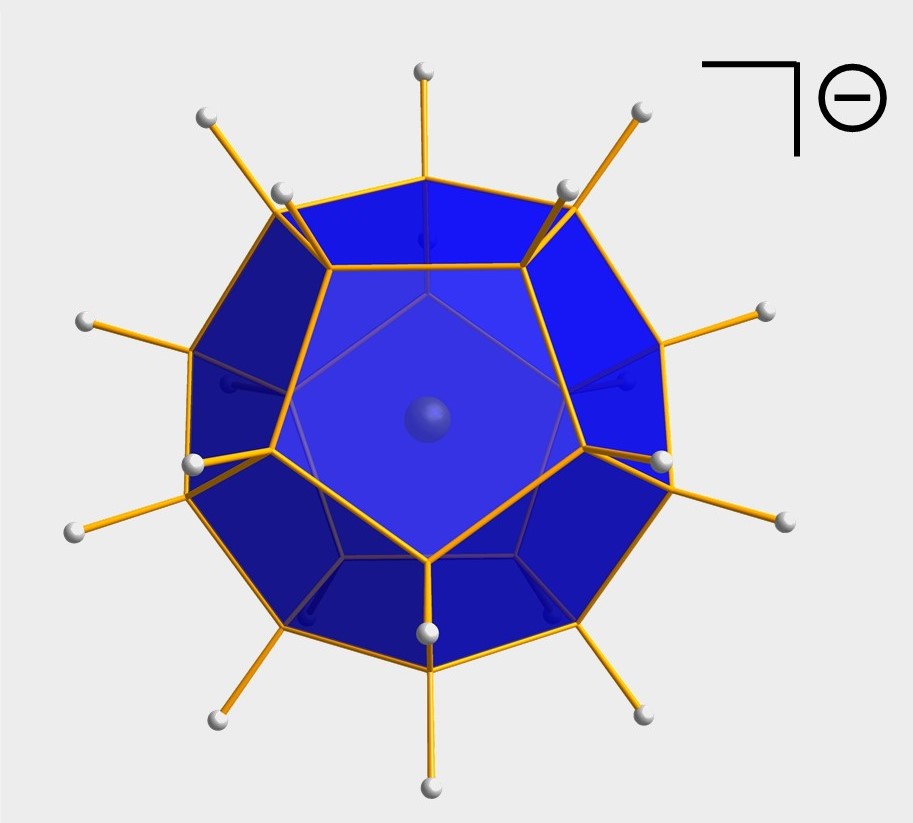 Organosilanes, oligosilanes, and oligo- germanes are indispensable precursors for key advanced materials, such as silicone polymers or Si/Ge semiconductors. We are therefore developing novel reactions to form bonds between group IV atoms (i.e., C-Si, Si-Si, Si-Ge, Ge-Ge) with the aim to create previously inaccessible molecular architectures. As an example, we have used the base-induced disproportionation of Si2Cl6 to conveniently assemble branched oligosilanes, cyclic oligosilanes, and even fullerene-type silicon nanocages (cf. Figure).
Organosilanes, oligosilanes, and oligo- germanes are indispensable precursors for key advanced materials, such as silicone polymers or Si/Ge semiconductors. We are therefore developing novel reactions to form bonds between group IV atoms (i.e., C-Si, Si-Si, Si-Ge, Ge-Ge) with the aim to create previously inaccessible molecular architectures. As an example, we have used the base-induced disproportionation of Si2Cl6 to conveniently assemble branched oligosilanes, cyclic oligosilanes, and even fullerene-type silicon nanocages (cf. Figure).